Key Findings from my PhD Dissertation:
Safety Conditioning is a therapeutic tool to reduce long-term contextual fear in a high-anxiety mouse strain.
I found that these behavioral improvements are followed by a decrease of perineuronal nets (PNNs) around parvalbumin (PV) interneurons within the infralimbic cortex (IL) & sustained PV activity at long-term memory retrieval.
I set out to determine how PNNs may regulate PV’s dynamic range & how PNNs are remodeled in a learning-specific manner. Through this work, I revealed a new non-myelinating role for oligodendrocytes in plasticity.
My work was the first to describe a learning-dependent role for satellite oligodendrocyte lineage cells (satOLCs).
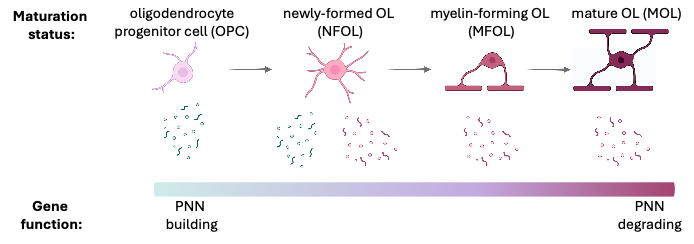
Using single-cell RNA sequencing data, I revealed that oligodendrocyte lineage cells (OLCs) differentially express genes that build or remodel the PNN.
OPCs express PNN structural genes.
Newly-formed OLs express both PNN structural genes & PNN degradation enzymes.
Mature OLs primarily express PNN degradation enzymes.
Proposed Outcome of satOL-PNN Regulation. Safety conditioning triggers satOLs to produce enzymes that degrade PNNs. This activity could have a range of effects, such as decreasing overall PNN density, remodeling of existing PNNs by increasing the size of holes (e.g., creating more real estate for new synaptic input), or preventing their formation, freeing PV from its constraints. With reduced PNN presence, PV INs are open to new synaptic connections, both activating and inhibitory. For example, GABAergic inputs would inhibit the PNN-free PV IN, disinhibiting local excitatory PNs (Wang et al., 2024). In this way, PV inhibitory activity is kept at an equilibrium, maintaining IL activation during fear inhibition. By limiting PNN expression, PV INs gain an expanded dynamic range, allowing them to activate selectively based on behavioral demands. This shift supports a balanced excitation-inhibition ratio, which in turn fosters adaptive learning, memory processes, and cognitive flexibility.